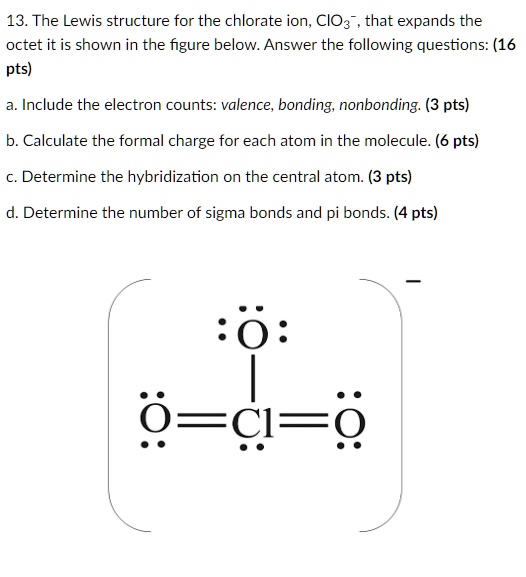
ClO3- lewis structure, molecular geometry, bond angle, hybridization | Molecular geometry, Molecular, Electron configuration

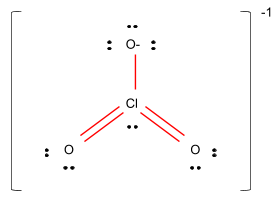
Based on formal charges, draw the most preferred lewis structure for the chlorate ion, ClO3−. - Chemistry QnA

How w ill ypu predict hybridisation of CLO3- show the steps - Chemistry - Chemical Bonding and Molecular Structure - 13429585 | Meritnation.com
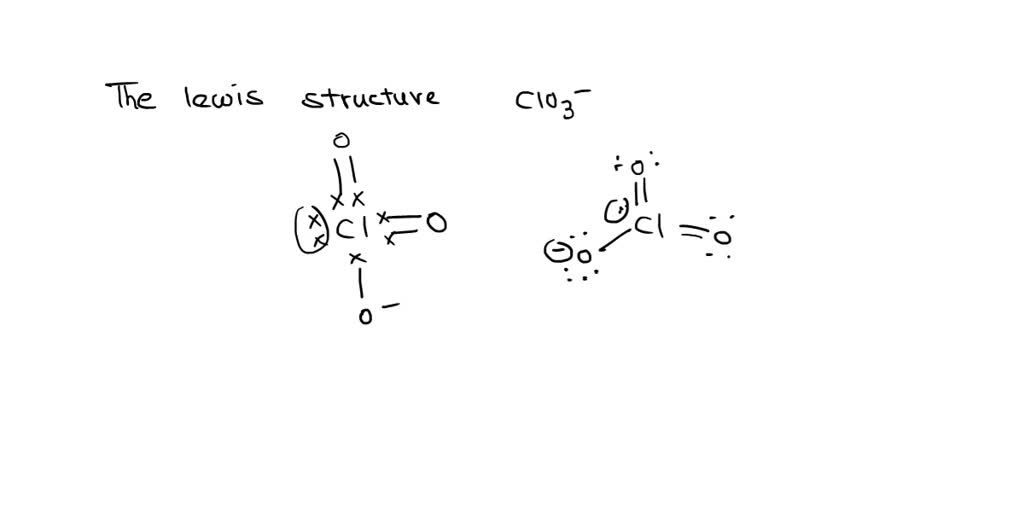
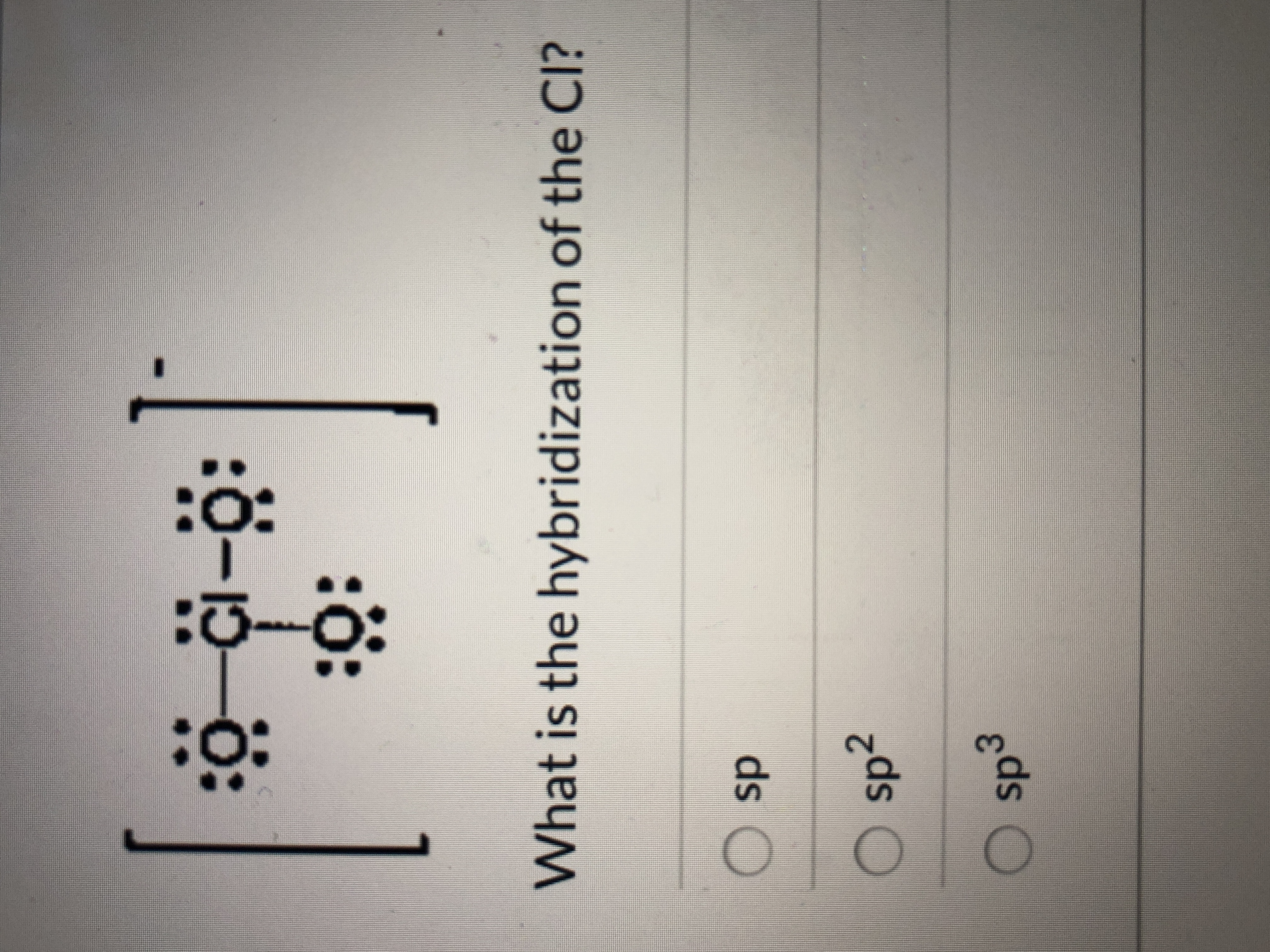
SOLVED: Draw the Lewis Structure of ClO3-. What is the hybridization of the central atom? sp3 sp3d sp2 sp3d2

Which of the following does not involve sp3hybridisation of the central atom ?A) H₃O+B) clo‐4C) SF4D) Clo3- - Brainly.in