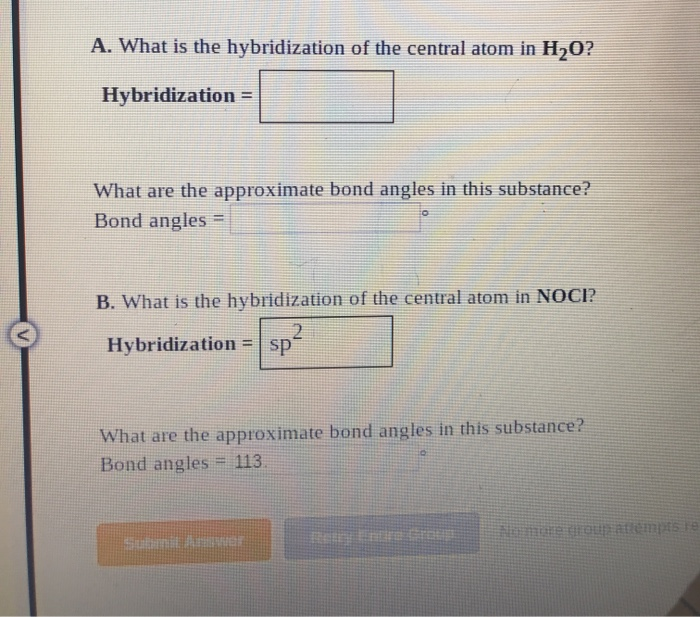
![The hybridisation and unpaired electrons in [Fe(H2O)6]2+ion are :a)sp3d2; 4b)d2sp3; 3c)d2sp3; 4d)sp3d2; 2Correct answer is option 'A'. Can you explain this answer? - EduRev Class 12 Question The hybridisation and unpaired electrons in [Fe(H2O)6]2+ion are :a)sp3d2; 4b)d2sp3; 3c)d2sp3; 4d)sp3d2; 2Correct answer is option 'A'. Can you explain this answer? - EduRev Class 12 Question](https://edurev.gumlet.io/ApplicationImages/Temp/3004871_40b115d7-9ff8-426c-8add-a9410d554b7d_lg.png)
The hybridisation and unpaired electrons in [Fe(H2O)6]2+ion are :a)sp3d2; 4b)d2sp3; 3c)d2sp3; 4d)sp3d2; 2Correct answer is option 'A'. Can you explain this answer? - EduRev Class 12 Question

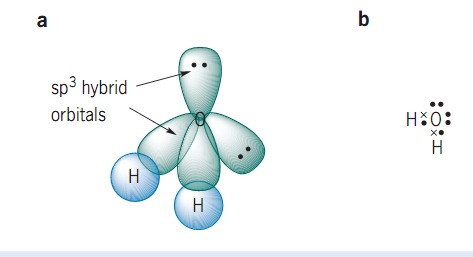
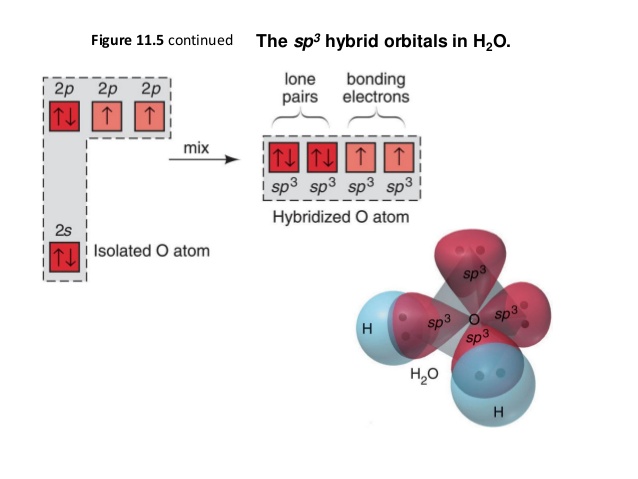
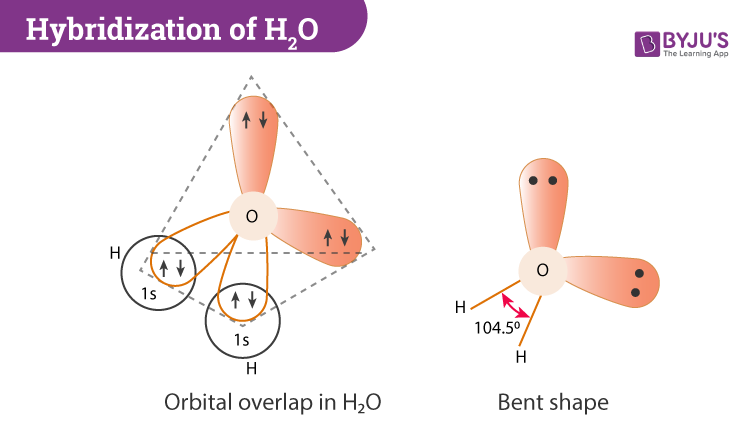
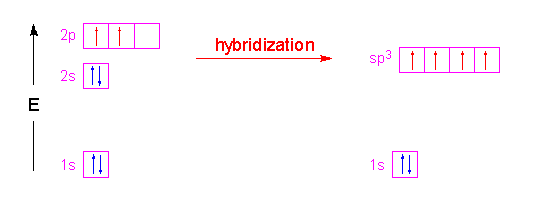
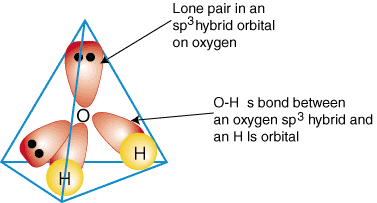
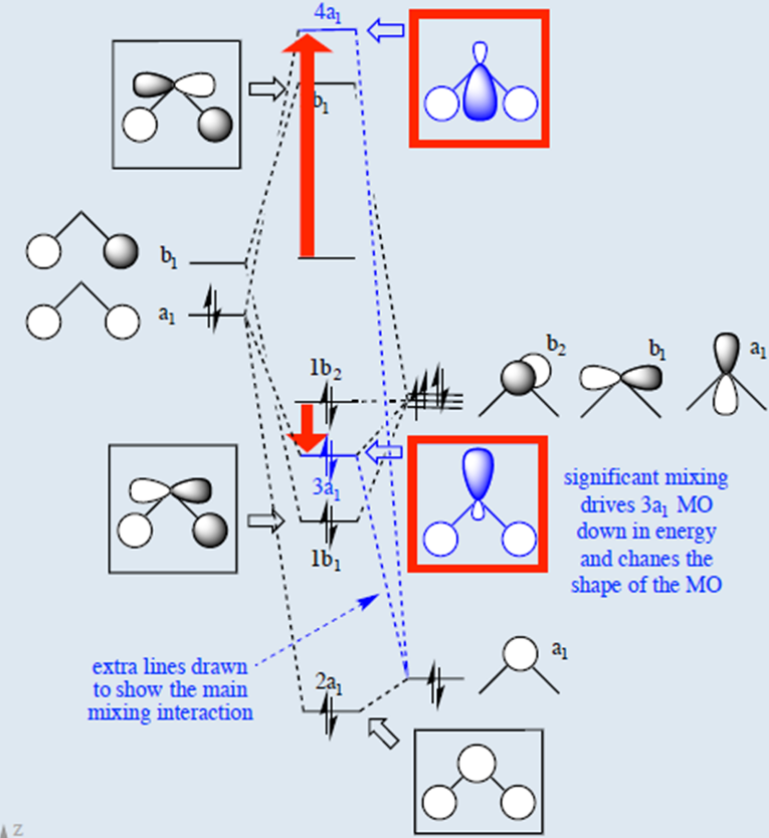
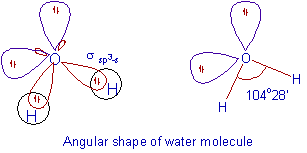
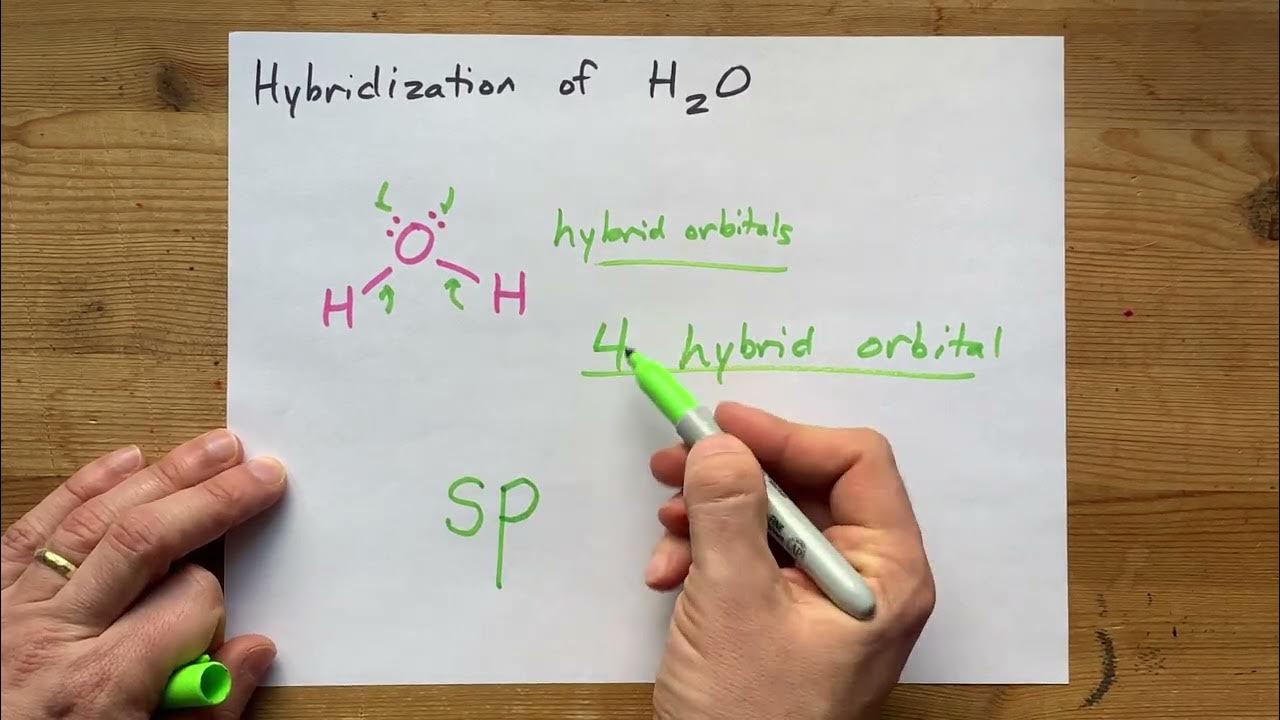
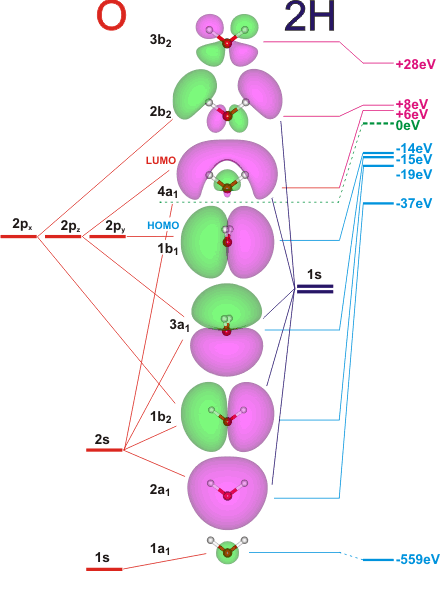
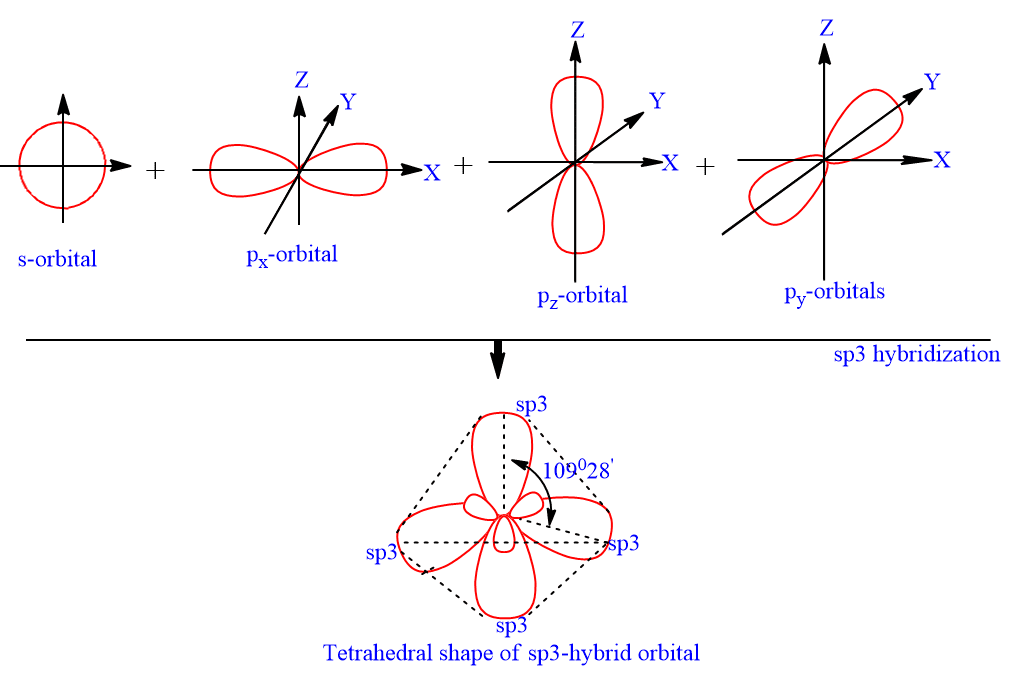
Please elaborate sp3 hybridisation in H2O molecule - Chemistry - Chemical Bonding and Molecular Structure - 13097221 | Meritnation.com