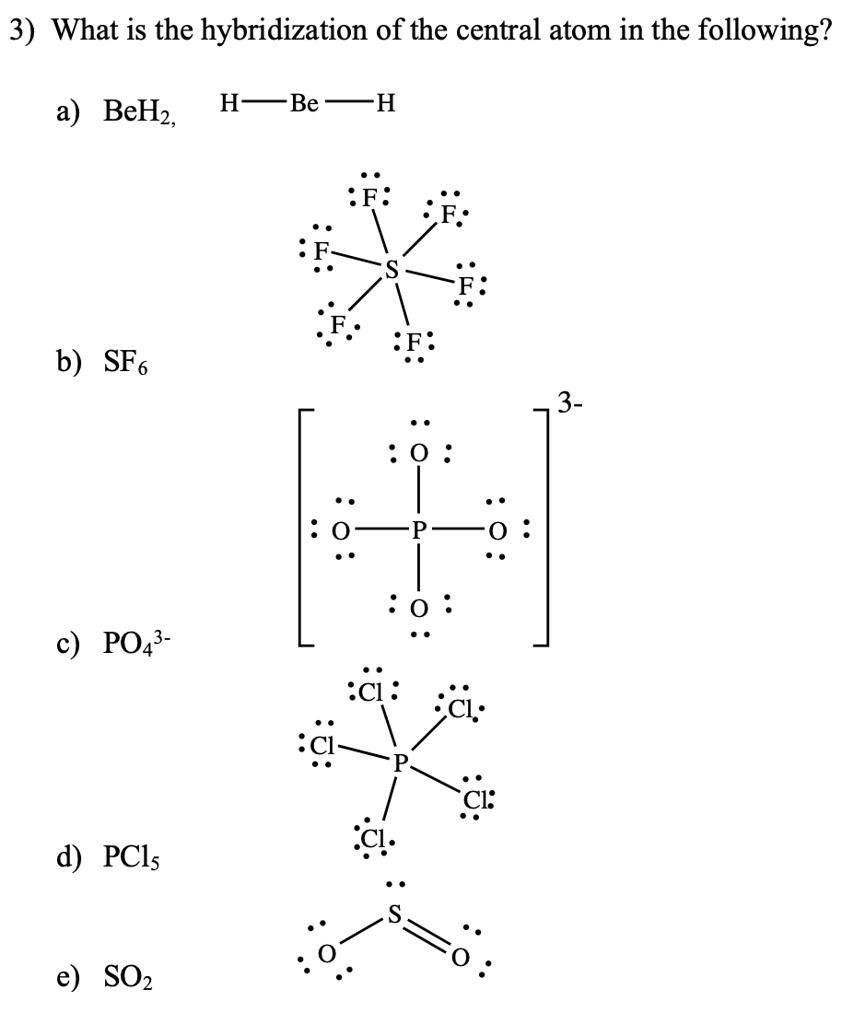
![Hybridisation of the underline atom changes in: [2002]a)AlH3 changes to AlH 4-b)H2O changes to H3O+c)NH3 changes to NH4+d)in all casesCorrect answer is option 'A'. Can you explain this answer? - EduRev JEE Hybridisation of the underline atom changes in: [2002]a)AlH3 changes to AlH 4-b)H2O changes to H3O+c)NH3 changes to NH4+d)in all casesCorrect answer is option 'A'. Can you explain this answer? - EduRev JEE](https://edurev.gumlet.io/ApplicationImages/Temp/634ddd4f-b583-4cd5-93a8-2591942ac108_lg.gif?w=360&dpr=2.6)
Hybridisation of the underline atom changes in: [2002]a)AlH3 changes to AlH 4-b)H2O changes to H3O+c)NH3 changes to NH4+d)in all casesCorrect answer is option 'A'. Can you explain this answer? - EduRev JEE

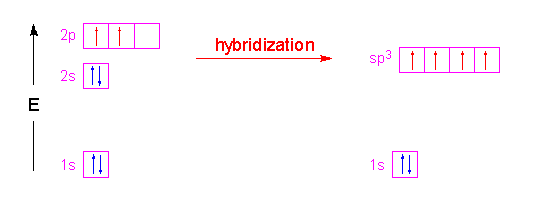
55. The hybridization of the central atom will change when 1) NHz combines with H+ 2) H,BOcombines with OH- 3) NHz forms NH, 4) H2O combines with H allowed. Use by non

In which one of the following species the central atom has the of hybridization which is not the same as that in the other three?

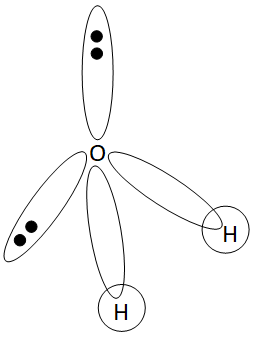
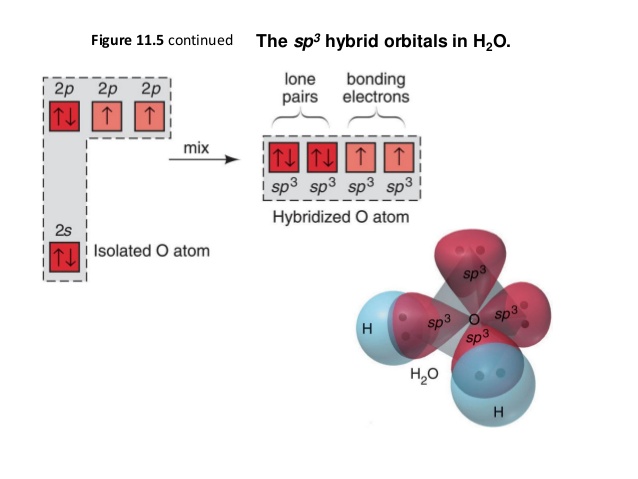
H2O Lewis Structure - Drawing Method of H2O Lewis Structure, Molecular Geometry of H2O, Polarity and Hybridisation in H2O molecule, with FAQs
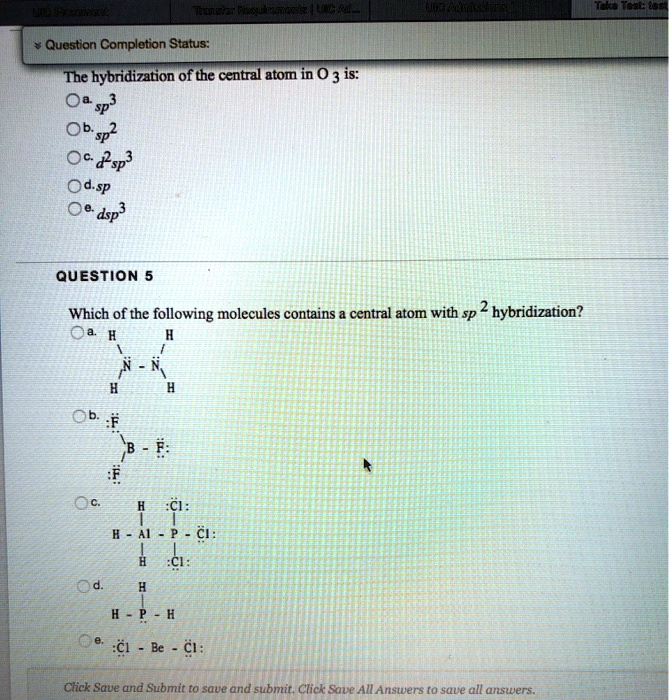
SOLVED: Q4L hybridization of the central atom in O3 is: dsp Which of the following molecules contains a central atom with sp2 hybridization? H2O Cl2 CO2