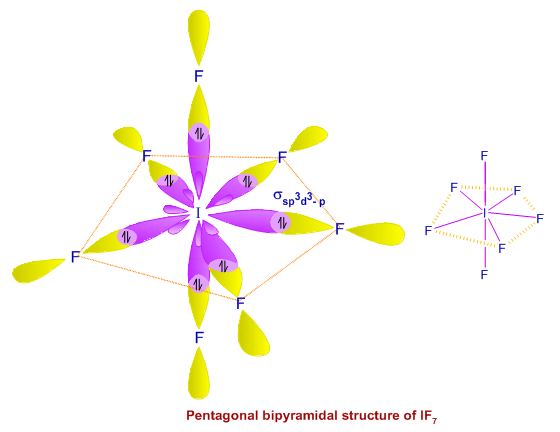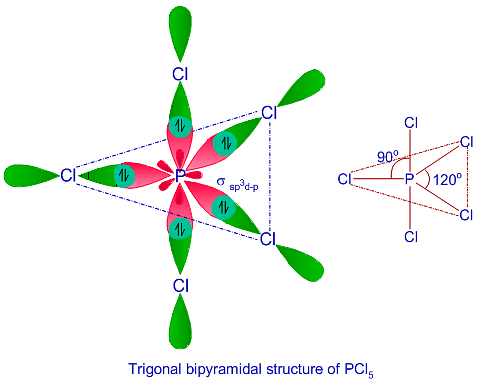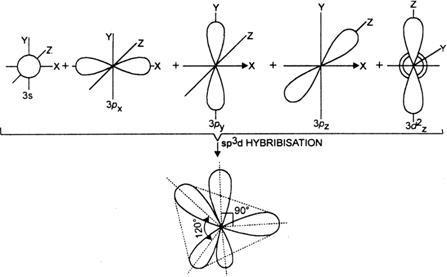
State and explain the geometric arrangements possible in sp3d and sp3d2 hybridisation. Name the d-orbitals involved in these. - Zigya
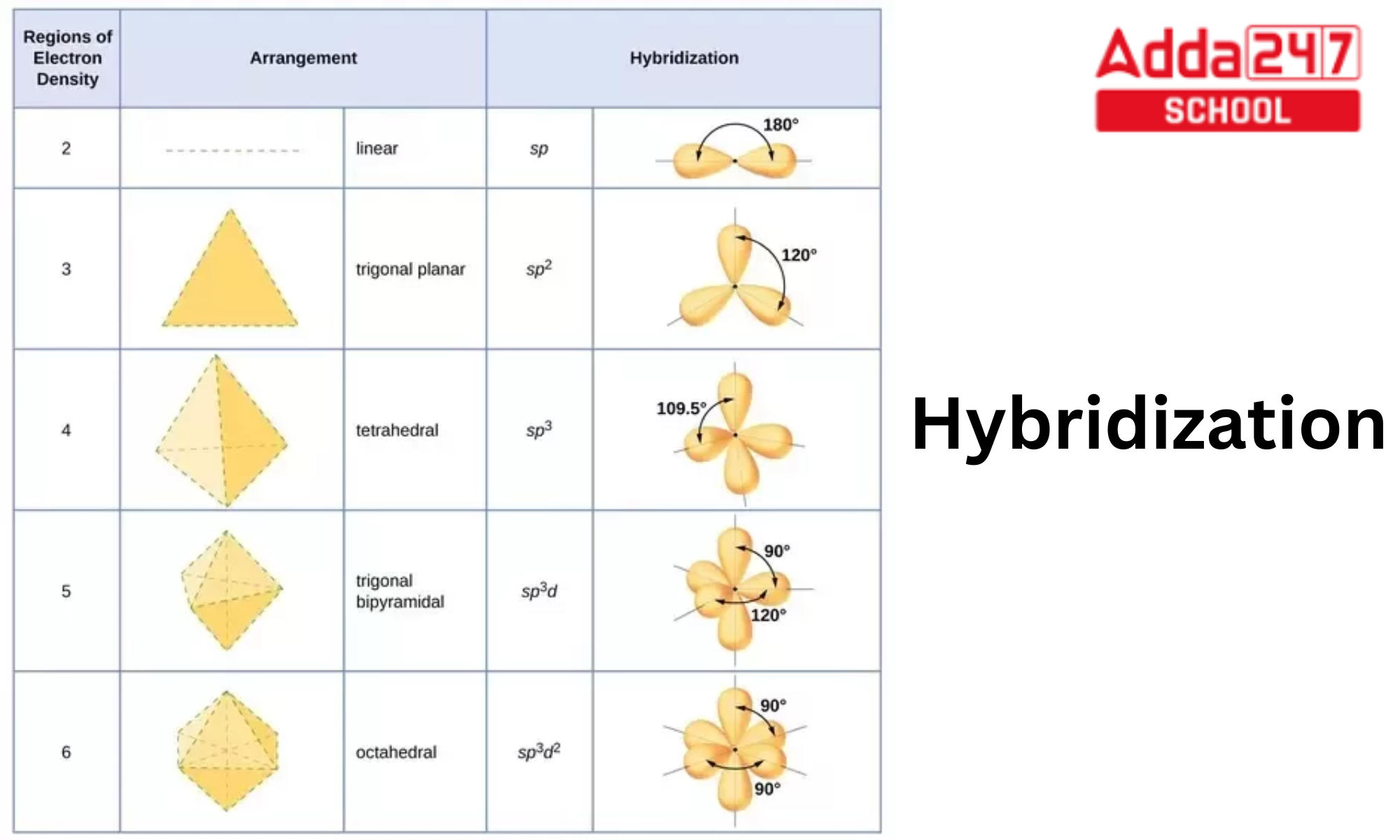
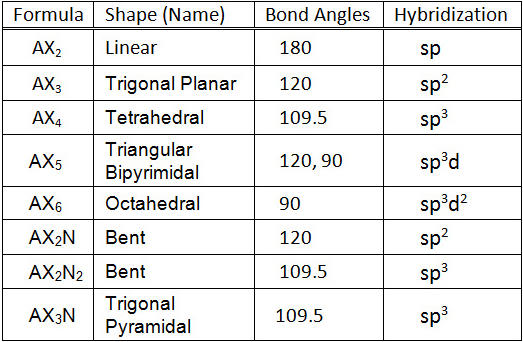
What are the shapes and bond angles of sp, sp2, sp3, sp3d, sp3d2 hybridised orbitals respectively? - Quora

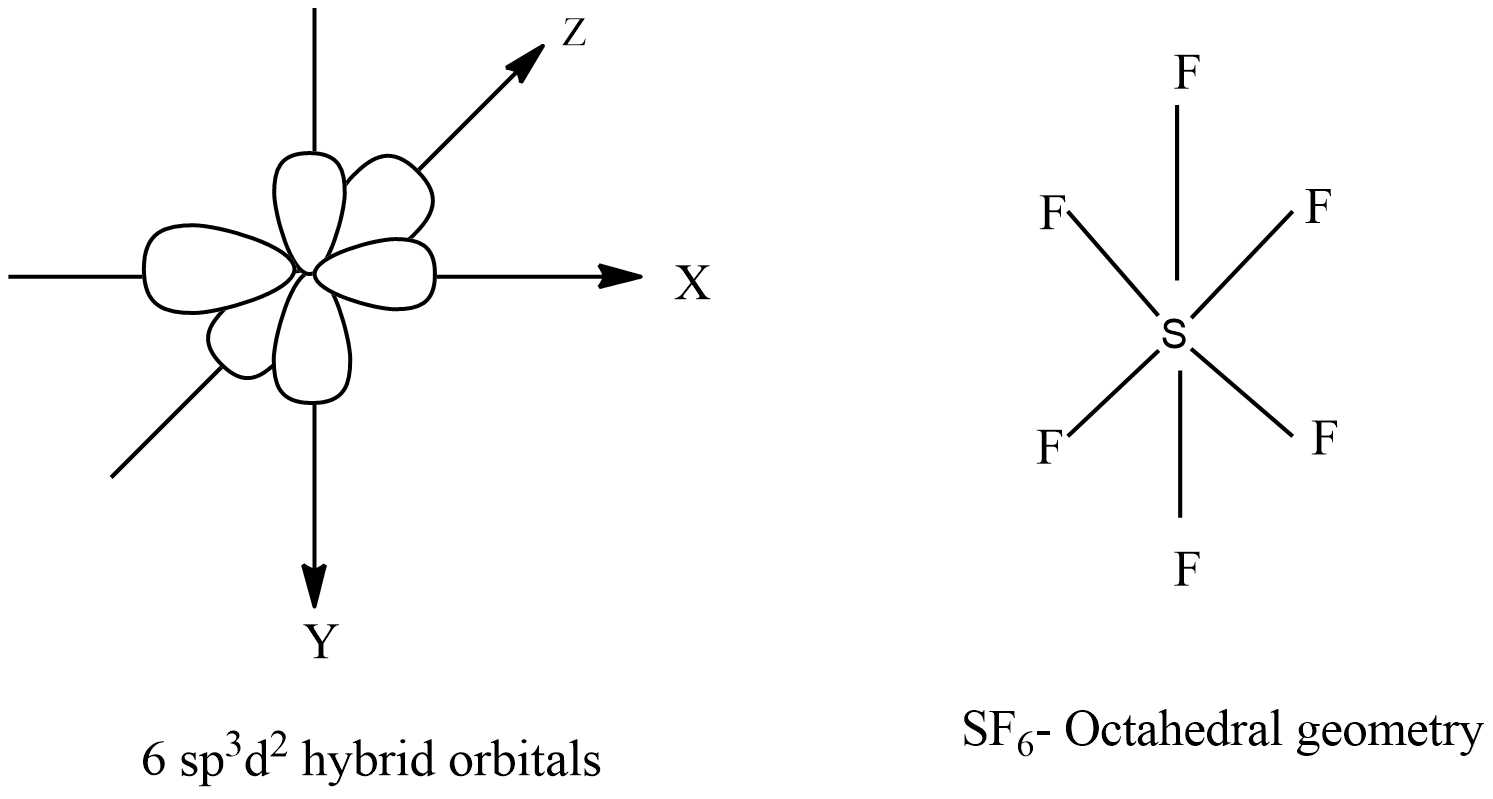
A molecule containing a central atom with sp3d2 hybridization has a(n) ______ electron geometry. a) trigonal planar b) trigonal bipyramidal c) octahedral d) tetrahedral e) trigonal pyramidal | Homework.Study.com
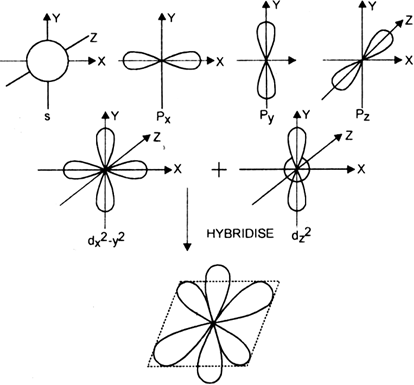
State and explain the geometric arrangements possible in sp3d and sp3d2 hybridisation. Name the d-orbitals involved in these. - Zigya
What are the shapes and bond angles of sp, sp2, sp3, sp3d, sp3d2 hybridised orbitals respectively? - Quora
What are the shapes and bond angles of sp, sp2, sp3, sp3d, sp3d2 hybridised orbitals respectively? - Quora
What are the shapes and bond angles of sp, sp2, sp3, sp3d, sp3d2 hybridised orbitals respectively? - Quora

Sp3d2 Hybridization Has 1s 3p And 2d Orbitals That Undergo Intermixing To Form 6 Identical Sp3d2 Hybrid Orbitals Stock Illustration - Download Image Now - iStock

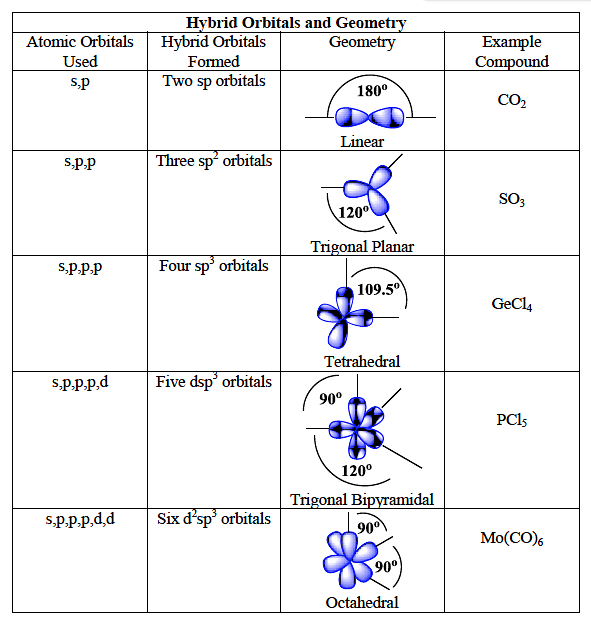
![Solved] Molecule obtained by sp3d2 hybridization has bond angle (s) Solved] Molecule obtained by sp3d2 hybridization has bond angle (s)](https://storage.googleapis.com/tb-img/production/21/08/Reported_23-Aug-2021_Shashi_D8.png)