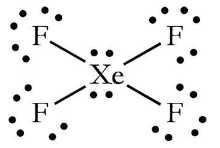
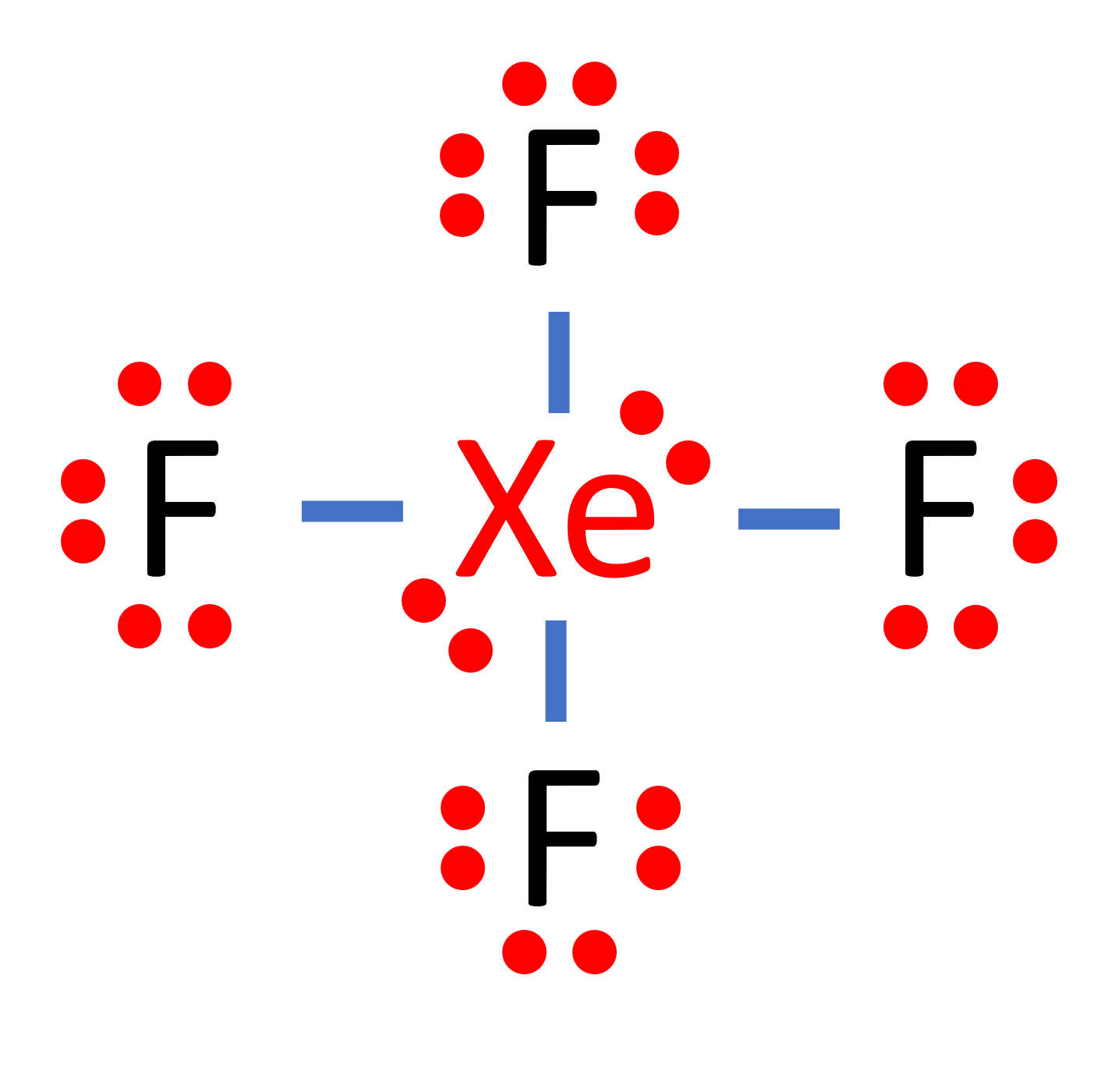
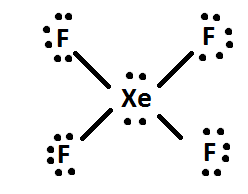
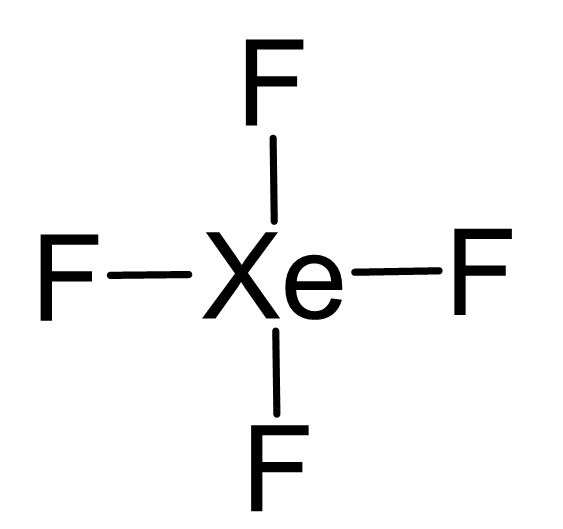87.The structure of XeF4 is square planer it could be bent as Co2 as in both cases there are 2 pairs of lone pair electron there was a possibility of lone pair

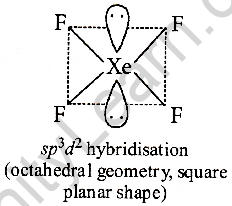
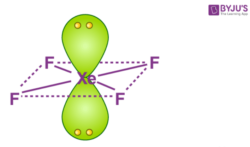
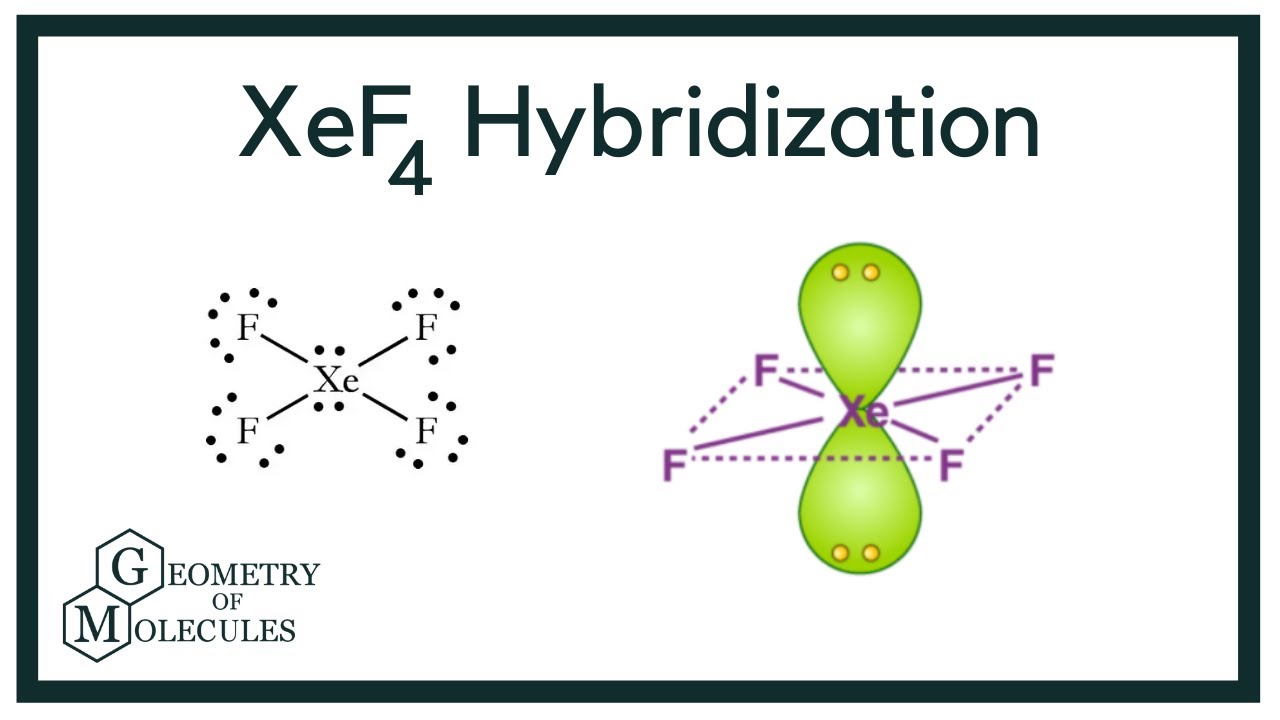
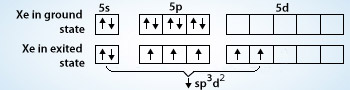
Chemistry Help on Instagram: "How is Xe atom hybridized in XeF4? Xenon tetrafluoride (XeF₄) is a compound that exhibits sp³d² hybridization. Hybridization is a concept in chemistry that describes the mixing of

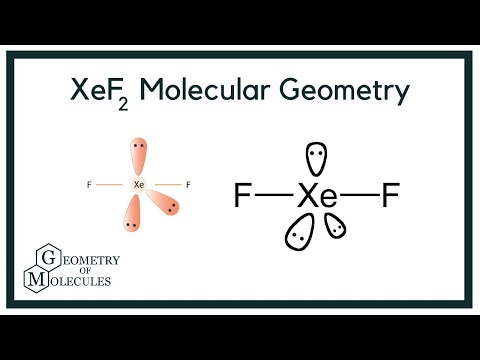
Hybridization of Xe in XeF4 is ........ and in XeF2 is ....... a. sp3, sp3d b. sp3d2, sp3d2 c. sp3d, sp3d2 d. sp3d2, sp3d e. sp3, sp3d2 | Homework.Study.com
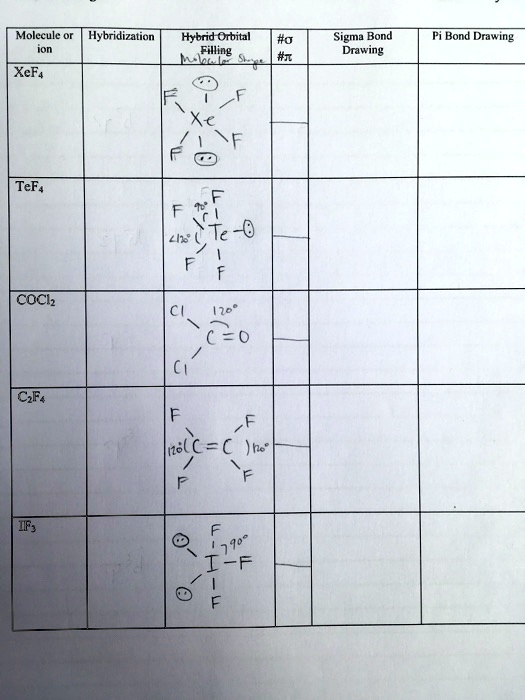
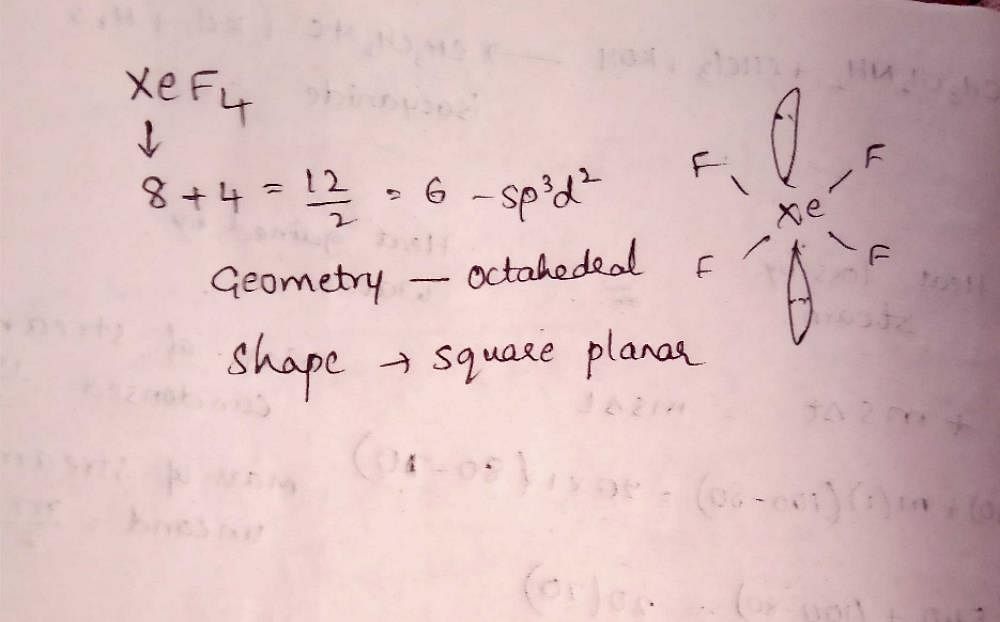




![ANSWERED] A. What is the hybridization of the central atom in XeF4 - Kunduz ANSWERED] A. What is the hybridization of the central atom in XeF4 - Kunduz](https://media.kunduz.com/media/sug-question/raw/52219992-1659248456.8030424.jpeg)